China Healthcare Reform and Its Impact on IVD, Medical Device and Pharmaceutical Markets
16 Nov 2011 • by Natalie Aster

New York — Private hospitals, high-end clinics and larger, better-funded public hospitals form the foundation of China’s urban healthcare market and China’s primary sales engine for many globally-leading medical product suppliers in the market.
The report “China Healthcare Reform and Its Impact on IVD, Medical Device and Pharmaceutical Markets” by Kalorama Information contains a comprehensive overview of the Chinese healthcare system with important data for marketers to China. It discusses healthcare reform plans and where funds were spent, and it describes the role foreign companies play.
Report Details:
China Healthcare Reform and Its Impact on IVD, Medical Device and Pharmaceutical Markets
Published: October 2011
Pages: 167
Price: US$ 3,500.00
Report Sample Abstract
Pharmaceutical industry investment in Chinese manufacturing by MNCs has evolved from packaging operations and encapsulation; to the production of active pharmaceutical ingredients (APIs) and intermediates; to the relocation of research and development (R&D) centers, laboratories and the launch of more intensive product manufacturing. Similarly, foreign medical device manufacturers have progressed beyond original equipment manufacturing (OEM) of device components and assembly of end-use products to the full production of medical equipment and technologies in China.
Foreign-Invested Manufacturing Enterprises in China (1980-2010)
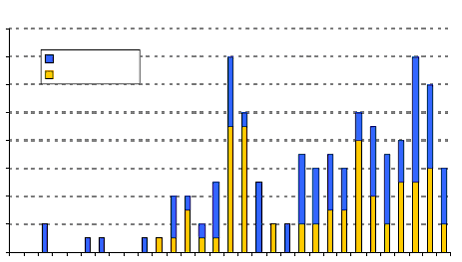
Foreign investment in comprehensive medical product manufacturing has complemented globally-significant domestic Chinese markets. China’s weight as a national market in the Asia/Pacific region and its untapped potential have also factored into MNC decisions to site R&D facilities in the country, which would be responsible for the development of products specifically for consumers in China and other developing Asian countries.
The regional contours and depth of investment in FIE manufacturing within China have been determined by numerous practical factors – logistics, incentives, historical development of SEZs and other areas open to FDI – but also several market forces.
More information can be found in the report “China Healthcare Reform and Its Impact on IVD, Medical Device and Pharmaceutical Markets” by Kalorama Information.
To order the report or ask for sample pages contact [email protected]
Contacts
MarketPublishers, Ltd.
Tanya Rezler
Tel: +44 208 144 6009
Fax: +44 207 900 3970
[email protected]
www.MarketPublishers.com
Analytics & News
