Familial Amyloid Neuropathies - Pipeline Review, H1 2020

SUMMARY
Global Markets Direct's latest Pharmaceutical and Healthcare disease pipeline guide Familial Amyloid Neuropathies - Pipeline Review, H1 2020, provides an overview of the Familial Amyloid Neuropathies (Metabolic Disorders) pipeline landscape.
Familial amyloid neuropathy is a slowly progressive condition characterized by the buildup of abnormal deposits of a protein called amyloid (amyloidosis) in the body's organs and tissues. Symptoms include numbness, tingling, pins and needles in the feet and hands, weakness and pain in the arms and legs, loss of sensation, urinary retention, reduced sweating, ankle swelling, fatigue, nausea, weight loss, dizziness and fainting. Supportive treatment includes diuretics, calcium channel blockers, beta blockers, angiotensin receptor blockers and angiotensin converting enzyme (ACE) inhibitors.
REPORT HIGHLIGHTS
Global Markets Direct's Pharmaceutical and Healthcare latest pipeline guide Familial Amyloid Neuropathies - Pipeline Review, H1 2020, provides comprehensive information on the therapeutics under development for Familial Amyloid Neuropathies (Metabolic Disorders), complete with analysis by stage of development, drug target, mechanism of action (MoA), route of administration (RoA) and molecule type. The guide covers the descriptive pharmacological action of the therapeutics, its complete research and development history and latest news and press releases.
The Familial Amyloid Neuropathies (Metabolic Disorders) pipeline guide also reviews of key players involved in therapeutic development for Familial Amyloid Neuropathies and features dormant and discontinued projects. The guide covers therapeutics under Development by Companies/Universities/Institutes, the molecules developed by Companies in Pre-Registration, Phase III, Phase II, Phase I, Preclinical and Discovery stages are 1, 2, 2, 2, 2 and 2 respectively. Similarly, the Universities portfolio in Phase II and Preclinical stages comprises 1 and 1 molecules, respectively.
Familial Amyloid Neuropathies (Metabolic Disorders) pipeline guide helps in identifying and tracking emerging players in the market and their portfolios, enhances decision making capabilities and helps to create effective counter strategies to gain competitive advantage. The guide is built using data and information sourced from Global Markets Direct’s proprietary databases, company/university websites, clinical trial registries, conferences, SEC filings, investor presentations and featured press releases from company/university sites and industry-specific third party sources. Additionally, various dynamic tracking processes ensure that the most recent developments are captured on a real time basis.
Note: Certain content/sections in the pipeline guide may be removed or altered based on the availability and relevance of data.
SCOPE
- The pipeline guide provides a snapshot of the global therapeutic landscape of Familial Amyloid Neuropathies (Metabolic Disorders).
- The pipeline guide reviews pipeline therapeutics for Familial Amyloid Neuropathies (Metabolic Disorders) by companies and universities/research institutes based on information derived from company and industry-specific sources.
- The pipeline guide covers pipeline products based on several stages of development ranging from pre-registration till discovery and undisclosed stages.
- The pipeline guide features descriptive drug profiles for the pipeline products which comprise, product description, descriptive licensing and collaboration details, R&D brief, MoA & other developmental activities.
- The pipeline guide reviews key companies involved in Familial Amyloid Neuropathies (Metabolic Disorders) therapeutics and enlists all their major and minor projects.
- The pipeline guide evaluates Familial Amyloid Neuropathies (Metabolic Disorders) therapeutics based on mechanism of action (MoA), drug target, route of administration (RoA) and molecule type.
- The pipeline guide encapsulates all the dormant and discontinued pipeline projects.
- The pipeline guide reviews latest news related to pipeline therapeutics for Familial Amyloid Neuropathies (Metabolic Disorders)
- Procure strategically important competitor information, analysis, and insights to formulate effective R&D strategies.
- Recognize emerging players with potentially strong product portfolio and create effective counter-strategies to gain competitive advantage.
- Find and recognize significant and varied types of therapeutics under development for Familial Amyloid Neuropathies (Metabolic Disorders).
- Classify potential new clients or partners in the target demographic.
- Develop tactical initiatives by understanding the focus areas of leading companies.
- Plan mergers and acquisitions meritoriously by identifying key players and it’s most promising pipeline therapeutics.
- Formulate corrective measures for pipeline projects by understanding Familial Amyloid Neuropathies (Metabolic Disorders) pipeline depth and focus of Indication therapeutics.
- Develop and design in-licensing and out-licensing strategies by identifying prospective partners with the most attractive projects to enhance and expand business potential and scope.
- Adjust the therapeutic portfolio by recognizing discontinued projects and understand from the know-how what drove them from pipeline.
Global Markets Direct Report Coverage
Familial Amyloid Neuropathies - Overview
Familial Amyloid Neuropathies - Therapeutics Development
Pipeline Overview
Pipeline by Companies
Pipeline by Universities/Institutes
Products under Development by Companies
Products under Development by Universities/Institutes
Familial Amyloid Neuropathies - Therapeutics Assessment
Assessment by Target
Assessment by Mechanism of Action
Assessment by Route of Administration
Assessment by Molecule Type
Familial Amyloid Neuropathies - Companies Involved in Therapeutics Development
ADRx Inc
Akcea Therapeutics Inc
Alnylam Pharmaceuticals Inc
BSIM Therapeutics SA
Corino Therapeutics Inc
Eidos Therapeutics Inc
Precision Biosciences Inc
Proclara Biosciences Inc
Prothena Corp Plc
Regeneron Pharmaceuticals Inc
Familial Amyloid Neuropathies - Drug Profiles
Antibody to Inhibit TTR for Familial Transthyretin Amyloidosis - Drug Profile
Product Description
Mechanism Of Action
R&D Progress
BBP-265 - Drug Profile
Product Description
Mechanism Of Action
R&D Progress
CLR-01 - Drug Profile
Product Description
Mechanism Of Action
R&D Progress
doxycycline hyclate - Drug Profile
Product Description
Mechanism Of Action
R&D Progress
eplontersen sodium - Drug Profile
Product Description
Mechanism Of Action
R&D Progress
Gene Therapy to Activate Transthyretin For Familial Amyloid Neuropathies - Drug Profile
Product Description
Mechanism Of Action
R&D Progress
NPT-189 - Drug Profile
Product Description
Mechanism Of Action
R&D Progress
NTLA-1001 - Drug Profile
Product Description
Mechanism Of Action
R&D Progress
patisiran - Drug Profile
Product Description
Mechanism Of Action
R&D Progress
PRX-004 - Drug Profile
Product Description
Mechanism Of Action
R&D Progress
Small Molecules to Inhibit Transthyretin Related Amyloid Protein for Familial Amyloidotic Polyneuropathy - Drug Profile
Product Description
Mechanism Of Action
R&D Progress
tolcapone - Drug Profile
Product Description
Mechanism Of Action
R&D Progress
vutrisiran - Drug Profile
Product Description
Mechanism Of Action
R&D Progress
Familial Amyloid Neuropathies - Dormant Projects
Familial Amyloid Neuropathies - Discontinued Products
Familial Amyloid Neuropathies - Product Development Milestones
Featured News & Press Releases
Dec 06, 2018: Eidos Therapeutics Appoints William Lis to Board of Directors
Nov 13, 2017: European Medicines Agency (EMA) Grants Alnylam Accelerated Assessment of Patisiran for Patients with Hereditary ATTR (hATTR) Amyloidosis
Nov 02, 2017: Sanofi and Alnylam Present Positive Complete Results from APOLLO Phase 3 study of Investigational Patisiran in Hereditary ATTR (hATTR) Amyloidosis Patients with Polyneuropathy
Sep 20, 2017: Arbutus’ LNP Licensee Alnylam Announces Positive Phase 3 Results for LNP-Enabled Patisiran Program
Sep 20, 2017: Sanofi and Alnylam Report Positive Topline Results from APOLLO Phase 3 Study of Patisiran in Hereditary ATTR (hATTR) Amyloidosis Patients with Polyneuropathy
Apr 26, 2017: Alnylam Reports Final 24-Month Results from Phase 2 Open-Label Extension Study of Patisiran, an Investigational RNAi Therapeutic in Development for the Treatment of Hereditary ATTR (hATTR) Amyloidosis
Oct 10, 2016: Data Monitoring Committee Recommends Continuation of APOLLO Phase 3 Clinical Trial of Patisiran for Hereditary ATTR Amyloidosis with Polyneuropathy (hATTR-PN)
Jul 01, 2016: Alnylam Reports New Results on patisiran at the XV International Symposium on Amyloidosis
Jun 27, 2016: Alnylam to Report New Patisiran Results at the XV International Symposium on Amyloidosis
Apr 20, 2016: Alnylam Reports Complete 18-Month Data from Ongoing Phase 2 Open-Label Extension Study of Patisiran, an Investigational RNAi Therapeutic Targeting Transthyretin for the Treatment of Hereditary ATTR Amyloidosis with Polyneuropathy (hATTR-PN)
Feb 01, 2016: Alnylam Completes Enrollment in APOLLO Phase 3 Study with Patisiran, an Investigational RNAi Therapeutic for Patients with Transthyretin (TTR)-Mediated Amyloidosis (ATTR Amyloidosis)
Nov 03, 2015: Alnylam Reports Positive Results from Ongoing Phase 2 Open-Label Extension Studies for Patisiran in Development for the Treatment of Transthyretin (TTR)-Mediated Amyloidosis (ATTR Amyloidosis)
Sep 28, 2015: Alnylam and Collaborators Report First-Ever Evidence that Patisiran Reduces Pathogenic, Misfolded Transthyretin Monomers and Oligomers in TTR-Mediated Amyloidosis Patients with Familial Amyloidotic Polyneuropathy
Jul 20, 2015: Alnylam Initiates Phase 3 Open Label Extension Study with Patisiran, an Investigational RNAi Therapeutic Targeting Transthyretin for the Treatment of TTR-Mediated Amyloidosis
Apr 21, 2015: Alnylam Reports 12-Month Clinical Data from Phase 2 Open Label Extension Study of Patisiran, an Investigational RNAi Therapeutic for Patients with Familial Amyloidotic Polyneuropathy
Appendix
Methodology
Coverage
Secondary Research
Primary Research
Expert Panel Validation
Contact Us
Disclaimer
Number of Products under Development for Familial Amyloid Neuropathies, H1 2020
Number of Products under Development by Companies, H1 2020
Number of Products under Development by Universities/Institutes, H1 2020
Products under Development by Companies, H1 2020
Products under Development by Universities/Institutes, H1 2020
Number of Products by Stage and Target, H1 2020
Number of Products by Stage and Mechanism of Action, H1 2020
Number of Products by Stage and Route of Administration, H1 2020
Number of Products by Stage and Molecule Type, H1 2020
Familial Amyloid Neuropathies - Pipeline by ADRx Inc, H1 2020
Familial Amyloid Neuropathies - Pipeline by Akcea Therapeutics Inc, H1 2020
Familial Amyloid Neuropathies - Pipeline by Alnylam Pharmaceuticals Inc, H1 2020
Familial Amyloid Neuropathies - Pipeline by BSIM Therapeutics SA, H1 2020
Familial Amyloid Neuropathies - Pipeline by Corino Therapeutics Inc, H1 2020
Familial Amyloid Neuropathies - Pipeline by Eidos Therapeutics Inc, H1 2020
Familial Amyloid Neuropathies - Pipeline by Precision Biosciences Inc, H1 2020
Familial Amyloid Neuropathies - Pipeline by Proclara Biosciences Inc, H1 2020
Familial Amyloid Neuropathies - Pipeline by Prothena Corp Plc, H1 2020
Familial Amyloid Neuropathies - Pipeline by Regeneron Pharmaceuticals Inc, H1 2020
Familial Amyloid Neuropathies - Dormant Projects, H1 2020
Familial Amyloid Neuropathies - Discontinued Products, H1 2020
Familial Amyloid Neuropathies is a slowly progressive condition characterized by the buildup of abnormal deposits of a protein called amyloid (amyloidosis) in the body's organs and tissues. Symptoms include numbness, tingling, pins and needles in the feet and hands, weakness and pain in the arms and legs, loss of sensation, urinary retention, reduced sweating, ankle swelling, fatigue, nausea, weight loss, dizziness and fainting. Supportive treatment includes diuretics, calcium channel blockers, beta blockers, angiotensin receptor blockers and angiotensin converting enzyme (ACE) inhibitors.
Familial Amyloid Neuropathies pipeline therapeutics constitutes close to 13 molecules.
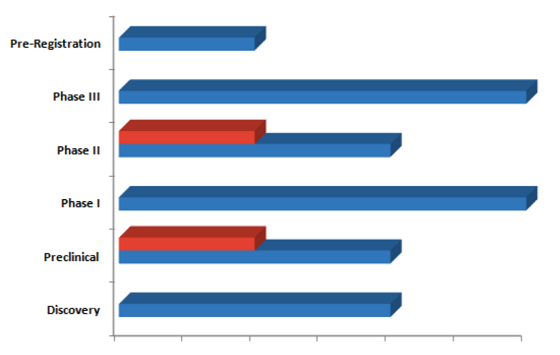
Global Markets Direct's latest report, 'Familial Amyloid Neuropathies - Pipeline Review, H1 2020', provides comprehensive information on the therapeutics under development for Familial Amyloid Neuropathies , complete with analysis by stage of development, drug target, mechanism of action (MoA), route of administration (RoA) and molecule type.

