Personalized Medicine Market - Advances in Human Genomics and Proteomics to Challenge Traditional Therapeutics
02 Dec 2010 • by Natalie Aster

The report “Personalized Medicine Market - Advances in Human Genomics and Proteomics to Challenge Traditional Therapeutics” by GBI Research provides an in-depth analysis on the potential applications of personalized medicine in pharmaceutical drug discovery and clinical services. The report suggests investment decisions in personalized medicine by providing information on valid personalized therapeutics, biomarkers, companion diagnostics, pipeline, the competitive landscape, mergers and acquisitions and market potential. In addition to this, the report also covers market drivers and restraints for the global personalized medicine market.
Report Details:
Published: December 2010
Pages: 124
Price: USD 3,500
Report Sample Abstract:
Companion Diagnostics to Foster the Transformational Trend Towards Personalized Medicine
Companion diagnostics facilitate identification of a patient's response to drugs depending on an individual’s genetic or proteomic profile. Companion diagnostics thus allow customization of treatment based on a patient's molecular or genetic make up, minimizing the risk of adverse reactions. Examples of successful therapeutics linked to companion diagnostic tests include Herceptin (trastuzumab), Gleevec (imatinib), Coumadin (warfarin), Camptosar (irinotecan) and Erbitux (cetuximab). Herceptin, for instance, is used to treat breast cancer patients that over-express HER2 protein. This subset of the patient population accounts for nearly 30% of the total breast cancer patients. HercepTest, the first test to be associated with a drug, was launched in 1998. HercepTest detects the over-expression of the HER2 protein, as a result increasing the accuracy and efficacy of treatment. Herceptin generated revenues of $5.2 billion in 2009, a growth of 10% from sales of $4.7 billion in 2007.
More and more companies are becoming increasingly involved in the development of companion diagnostics owing to the cost-effectiveness and accuracy of companion diagnostics in treatment and the drug discovery process as well. Oncology, in particular, is witnessing increased acceptance of companion diagnostics as the conventional trial and error method can prove to be cumbersome in patients with standard care. Companion diagnostics is also referred to as theranostics, holds immense potential, raising the prospect of a new standard of healthcare.
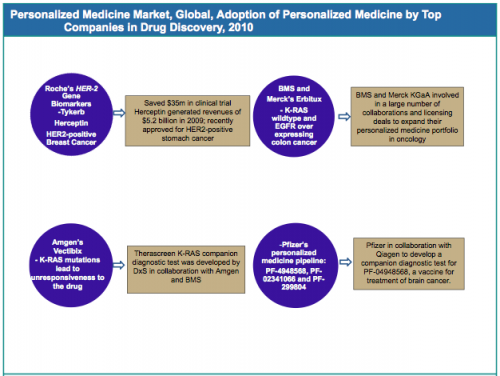
Major Players Are Becoming Increasingly Aligned Around Personalized Medicine – Oncology Continues to be the Prime Focus
Developing a new drug is a costly and time-consuming process. The application of advanced technologies for the development of personalized medicine, be it diagnostics or therapeutics, in the drug discovery process is becoming increasingly evident. The integration of pharmacogenomic data in clinical trials and drug development process helps in selecting patients that are more likely to respond or least likely to suffer any side effects. This considerably reduces the time, size and expense of clinical trials and also helps pharmaceutical companies to overcome the challenges posed by conventional processes which are time consuming and expensive.
Application of personalized medicine in the pharmaceuticals sector has the potential to revolutionize drug discovery process by facilitating treatment decisions and introducing preventive care. Applications of personalized medicine in oncology are becoming increasingly prominent, owing to genetic variations causing tumors and the resulting genetically defined patient subtypes. A number of therapeutics and biomarkers are focused on cancer research to allow better understanding of the disease. Within oncology, the major indications are breast cancer, lung cancer and colorectal cancer. The other major therapeutic areas include infectious diseases, immunology and cardiovascular diseases.
Source: GBI Research, Company Websites, US FDA (United States’ Food and Drug Administration)
Fundamental Business Models Including Regulatory and Reimbursement Regimes Need to be Revisited
Regulatory approval of innovative diagnostics or companion diagnostics will require a new regulatory process that considers the relevance of individual genetic profiles of patients and clinical outcomes. In the US, the Food and Drug Administration (FDA) and the Center for Medicare and Medicaid Services (CMS) are authorized to regulate and oversee the approval of medical devices, diagnostics and therapeutics and compliance with performance standards. The FDA is very supportive of the advancements in personalized medicine. Approval of certain genetic assays, reviews of pharmacogenomic guidance for the industry and the publication of papers and reviews on various related concepts such as biomarkers and drug-diagnostic co-development are some of the many efforts taken by the FDA in encouraging public education in personalized medicine. However, clear regulatory guidelines on the requirements for the development of companion diagnostics are yet to be formulated.
The success of personalized medicine relies largely on adequate payer coverage and reimbursement. Increasing evidence from pharmacogenomics research suggests that the conventional “one drug fits all” model doesn’t hold true in all cases. Thus, treatment has to be customized based on individual genetic profiles. Reimbursement policies, particularly for companion diagnostic tests, need to be realigned to keep pace with scientific progress and allow for increased adoption by physicians and patients.
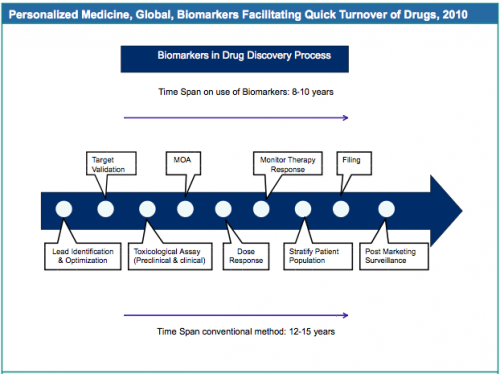
Biomarkers Facilitate Quick Turnover of Drugs and Significant Reduction in Drug Discovery and Development Costs
High drug attrition rates, safety and efficacy concerns, and time consuming methods, have encouraged the pharmaceuticals sector to adopt the use of biomarkers in the drug discovery process on a large scale. According to the FDA the biomarkers are 10% more efficient in predicting drug failures and as a result can save approximately $100m and three to four years in each drug discovery process. In addition, drugs that are failing in clinical trials or have been withdrawn from the market due to unresponsiveness of a broader patient population can be rescued and reconsidered for studies in benefiting a subset of population.
Source: GBI Research, FDA
More information can be found in the report “Personalized Medicine Market - Advances in Human Genomics and Proteomics to Challenge Traditional Therapeutics ” by GBI Research.
To order the report or ask for sample pages contact [email protected]
Contacts
MarketPublishers, Ltd.
Mrs. Alla Martin
Tel: +357 96 030922
Fax: +44 207 900 3970
[email protected]
www.marketpublishers.com
Analytics & News
