CE Dietary Supplements Market to Grow by 7.6% Annually through 2014, Forecasts PMR
06 Mar 2013 • by Natalie Aster

In 2012 the market growth rate for the dietary supplement pharmacy market in the CE region slowed to 7%, but the market exceeded €1bn for the first time in history. In all, the CAGR (compound annual growth rate) for 2012-2014 will be 7.6%, according to the report “Dietary Supplements Market in Central Europe 2012” by PMR.
Report Details:
Dietary Supplements Market in Central Europe 2012
Published: January, 2013
Pages: 227
Price: US$ 3,500.00
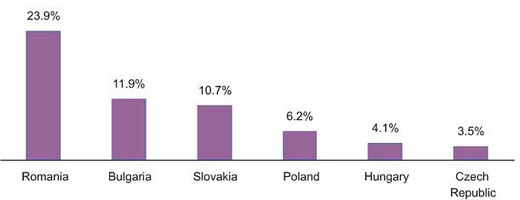
According to PMR estimates, in 2010 and 2011 the dietary supplement market in Romania increased more than in other countries, by almost 24% per annum on average. There was only a slight increase, of around 3.5%, in the dietary supplement market in the Czech Republic (because the market in this country is the most saturated). In 2011 the Czech Republic had the highest figure for per capita spending on dietary supplements (at pharmacies), among the CE countries analysed. Romania was at the other end of the scale. “The situation will not have changed significantly by 2014. The Czech Republic will remain the most highly developed dietary supplement market in the region. The most attractive market in the next three years in the region, among the countries analysed, taking forecast market growth into account, will be Romania” says Agnieszka Skonieczna, PMR Senior Pharmaceutical Market Analyst and the report co-author.
Average CAGR for Dietary Supplement Markets in the Analysed CE Countries (%), 2010-2011
Will new health claims regulations fail to revolutionise the dietary supplement market?
On 14th December 2012, EU Regulation No. 432/2012 came into force. This defines the list of permitted health claims pertaining to foods, other than statements relating to a reduction in disease risk, development and health. Packaging and advertising of dietary supplements will only be able to mention health statements found on the EU “white list”, which according to lawyers will severely restrict their advertising. There are only 222 health claims found on the “white list” and over 1,700 have been rejected and cannot be used. 14th December 2012 is also the date on which the transition period ended – after that date, labels, advertisements, websites, leaflets and all other forms of presentation and advertising had to be free of all banned content.
“According to companies interviewed by PMR specifically for the purposes of the report, new health claims regulations will not have as substantial an effect on the dietary supplement market as would have been expected” says Monika Stefanczyk, PMR Head Pharmaceutical Market Analyst and the report co-author. For example, the regulation does not affect products which have herb-based active ingredients, and there are many such products on the Hungarian market (including vitamins combined with herbs), which still can be sold with the old labelling. In Bulgaria companies believe that there will be some initial difficulty before the new requirements are met but that the change will be positive for the industry in the long term. Most of the changes will affect small producers and should lead to a reduction in unfair competition. Although the companies need to ensure very quickly that their products comply with the new regulations, our respondents in Slovakia believe that most companies active on the market will do this and that few products will disappear.
Mergers and acquisitions on the CE dietary supplement market
2011 and 2012 brought a number of mergers and acquisitions on the dietary supplement market in Central Europe. In Poland, Laboratoria Natury, a Polish producer of dietary supplements, agreed to be acquired by Maabarot Products, an Israeli manufacturer of nutritional supplements and food for people and animals. Herbapol-Lublin, a producer of herbal OTC medications and dietary supplements, acquired shares in Bio-Active, which specialises in the sale of teas which have the status of dietary supplements, and Herbapol Pruszkow, another herbal product manufacturer. PharmaSwiss, a subsidiary of Valeant Pharmaceuticals, acquired certain brands belonging to VitaDirect, a Warsaw-based maker of dietary supplements, pharmaceutical cosmetics and medicinal products.
Mid Europa Partners, a private equity fund, agreed to buy a 50% stake in Walmark, a prominent Czech dietary supplement manufacturer. Labormed, a prominent Romanian manufacturer, sold part of its portfolio to Recordati, an Italian drug producer.
More information can be found in the report “Dietary Supplements Market in Central Europe 2012” by PMR.
To order the report or ask for sample pages contact [email protected]
Contacts
MarketPublishers, Ltd.
Tanya Rezler
Tel: +357 96 030922
Fax: +44 207 900 3970
[email protected]
MarketPublishers.com
Analytics & News
