Aspergillosis Therapeutics Market Forecast to Grow by 1.5% in Foreseeable Future, Reports GlobalData
31 Jul 2012 • by Natalie Aster

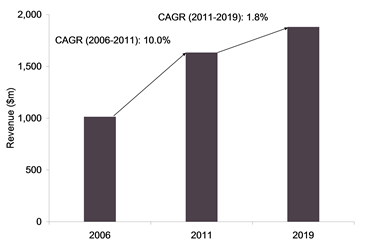
The global aspergillosis therapeutics market was worth $1.6 billion in 2011, and is forecast to grow at a CAGR of 1.5% for the next eight years, to reach $1.9 billion by 2019. The aspergillosis therapeutics market is formed primarily by four classes of antifungal drugs: polyenes, triazoles, echinocandins and allylamines. The most prescribed drugs for the treatment of aspergillosis are Vfend, Noxafil, Cancidas, itraconazole and amphotericin B, of which the latter two are generics. Vfend is the first line of therapy and the gold standard of treatment for aspergillosis.
GlobalData’s new report “Aspergillosis Therapeutics - Pipeline Assessment and Market Forecasts to 2019” suggests that the global aspergillosis therapeutics market is showing slow growth due to the expiry of the patents for the major marketed drugs. Cancidas’ patent will expire in 2015, followed by the expiry of the patents for Vfend and Noxafil in 2015 and 2019 respectively. However, market growth will be augmented by the increasing incidence rate and the expected launch of a new triazole, Isavuconazole. Isavuconazole’s launch will not have a significant impact on the aspergillosis therapeutics market as it is a me-too product, and faces competition with the generic triazole drugs.
The report is an essential source of information and analysis on the global aspergillosis therapeutics market. The report identifies the key trends shaping and driving the global aspergillosis therapeutics market. The report also provides insights on the prevalent competitive landscape and the emerging players expected to significantly alter the market positioning of the current market leaders. Most importantly, the report provides valuable insights on the pipeline products within the global aspergillosis therapeutics sector. This report is built using data and information sourced from proprietary databases, primary and secondary research and in-house analysis by GlobalData’s team of industry experts.
Aspergillosis Therapeutics, Global, Revenue and Market Forecast ($m), 2006–2019
GlobalData has found that the global aspergillosis therapeutics market holds potential, as the unmet need within it is significant. There are currently four classes of antifungal drug prescribed for the treatment of aspergillosis. The currently approved products moderately serve the aspergillosis therapeutics market, in which the mortality and morbidity rates are high. The current available drugs are moderately efficacious in increasing patients’ survival rate, but show a poor safety profile – they are associated with high toxicity and adverse drug interaction. The cure rate in patients with these drugs is also low. Patient compliance with the therapy is also a concern – antifungal therapy continues for a long period of time and Vfend is the only approved drug in oral formulation. A significant opportunity exists in the aspergillosis therapeutics market for the new entrants. An efficacious product with a high safety profile will have the potential to capture a significant share of the market. The development of antifungal drugs in oral formulation will improve patient compliance.
Report Details:
Aspergillosis Therapeutics - Pipeline Assessment and Market Forecasts to 2019
Published: March, 2012
Pages: 63
Price: US$ 3.596,00
GlobalData’s analysis of the aspergillosis pipeline has found it to be weak, as it lacks novel molecules in the late stage of development. The aspergillosis pipeline consists of 11 molecules in different stages of development. The late stage pipeline (Phase III) consists of two me-too molecules, Isavuconazole and SPK-843 which are triazole and polyene antifungals respectively. Isavuconazole is being developed by Astellas in association with Basilea Pharmaceutica and is expected to be launched in 2014.
One more product extension molecule, a combination of voriconazole and anidulafungin, was in development, but the latest Phase III trials have shown that the combination was not able to show statistically significant results in comparison to voriconazole alone. As the current aspergillosis therapeutics market is well served by recently approved antifungal drugs; these late stage molecules will face strong competition from the existing approved molecules, when approved.
More information can be found in the report “Aspergillosis Therapeutics - Pipeline Assessment and Market Forecasts to 2019” by GlobalData.
To order the report or ask for sample pages contact [email protected]
Contacts
MarketPublishers, Ltd.
Tanya Rezler
Tel: +44 208 144 6009
Fax: +44 207 900 3970
[email protected]
MarketPublishers.com
Analytics & News
