The Cardiovascular Market Outlook to 2016
14 Jul 2011 • by Natalie Aster
“The large number of impending patent expirations of blockbuster drugs will negatively impact the cardiovascular pharmaceutical segment. Evolving strategies in this segment such as in-licensing of drug candidates, and marketing of authorized generics, along with the development of first-in-line novel therapeutics might offset the effects of genericization.”
The report “The Cardiovascular Market Outlook to 2016” by Business Insights provides an overview of the cardiovascular market, including analyses of the disease subcategories, market sizing, market forecasts, and growth drivers.
Cardiovascular diseases encompass a range of ailments such as hypertension, dyslipidemia, stroke, atherosclerosis, thrombosis, and coronary artery disease. Dyslipidemia was the indication with the highest reported prevalence in 2010 with an estimated 336 million cases in the seven major markets.
Antidyslipidemic drugs occupied 18% of the cardiovascular segment in 2010. The entry of Lipitor generics in 2011 is expected to negatively impact future prospects of sales in this segment. Dalcetrapib, a CETP inhibitor, manufactured by Roche, is a potential blockbuster in this segment that could offset the negative impact of genericization.
Antithrombotics recorded strong sales in 2010, and occupied a market share of 11%. This segment is forecast to deliver strong growth in the next two years owing to the commercialization of recently approved products such as Pradaxa and Xarelto and the expected launch of Pfizer/BMS’s apixaban in the US.
Report Details:
The Cardiovascular Market Outlook to 2016
Published: June 2011
Pages: 160
Price: US$ 3,835.00
Report Sample Abstract
The antithrombotic category is characterized by a relatively strong developmental pipeline, with several products that could prove to be the future blockbusters. Most of these drugs are anticoagulants, which are aimed at replacing warfarin. Together, they could exhibit potential sales of about $11-12bn in 2021.
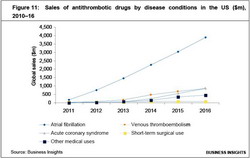
Among the various disease conditions, atrial fibrillation shows the greatest potential for sales of the new antithrombotics (as shown in Figure 11). These drugs are likely to capture significant market share from Lovenox, which will lose patent exclusivity by 2012.
Currently only 30% of atrial fibrillation conditions are treated with warfarin. Thus, stress would be laid on those antithrombotic drugs with lower bleeding risks than warfarin, as these could be used to treat atrial fibrillation.
Diagnosis, treatment and management
In over 90% of cases there may be no identifiable cause of hypertension, in which case the condition is known as essential hypertension. Some researchers believe that this may be due to hormonal factors, which control the salt-handling ability of the kidney, while others believe that it is genetically determined and environmentally controlled. In the remaining 10% of cases, hypertension may be secondary in nature as a consequence of another medical problem, such as kidney disorders, adrenal tumors, or some drugs.
Key events in the cardiovascular market
European approval for Rasilamlo
Rasilamlo, a single pill combination of a rennin inhibitor (aliskiren) and a calcium channel blocker (amlodipine), manufactured by Novartis, received approval from the European Commission in April 2011 for the treatment of high blood pressure in patients who are not controlled by either one of the antihypertensive molecules. The approval is based on studies in more than 5,000 patients with mild to severe blood pressure, and establishes the superiority of the combined therapy over the use of a single therapy. Novartis also filed a request for EU approval for a triple combination product in May 2010. The triple combination product, Amturnide, consists of aliskiren, amlodipine, and hydrochlorothiazide (a diuretic).
More information can be found in the report “The Cardiovascular Market Outlook to 2016” by Business Insights.
To order the report or ask for sample pages contact [email protected]
Contacts
MarketPublishers, Ltd.
Tanya Rezler
Tel: +357 96 030922
Fax: +44 207 900 3970
Analytics & News
